
It has been proposed that ZP proteins self-aggregate into fibrillar structures via cross-β-sheets, similar to the structure of amyloids. Although the ZP and somatic cell ECM consist of different proteins, they have certain properties in common, such as viscoelasticity that can affect cellular behavior. Mutations in ZPD genes can result in severe human pathologies such as vascular disease, renal disease, deafness, cancer, or infertility. Subdomain ZP-N is involved in polymerization of nascent ZP proteins into fibrils, as well as in polymerization of many other ZPD-containing proteins, such as tectorin, uromodulin, mesoglein, and cuticlins, into fibrils and matrices. These subdomains adopt immunoglobulin (Ig)-like folds and are connected to each other by a short, protease-sensitive linker region. The latter mutations provide additional support for the role of ZPD subdomains and other regions of ZP polypeptide in polymerization of human ZP proteins into fibrils and matrix.Įach ZP protein has a zona pellucida domain (ZPD) that consists of ≈270 amino acids (aa), 8 or 10 conserved cysteine (Cys) residues present as intramolecular disulfides, and two subdomains, ZP-N and ZP-C. Gene sequence variations in human ZP genes due to point, missense, or frameshift mutations also have a detrimental effect on ZP formation and female fertility. Inactivation of mouse ZP genes by gene targeting has a detrimental effect on ZP formation around growing oocytes and female fertility. The presence of both ZP2 and ZP3 is required to assemble ZP fibrils and ZP1 and ZP4 are used to crosslink the fibrils. 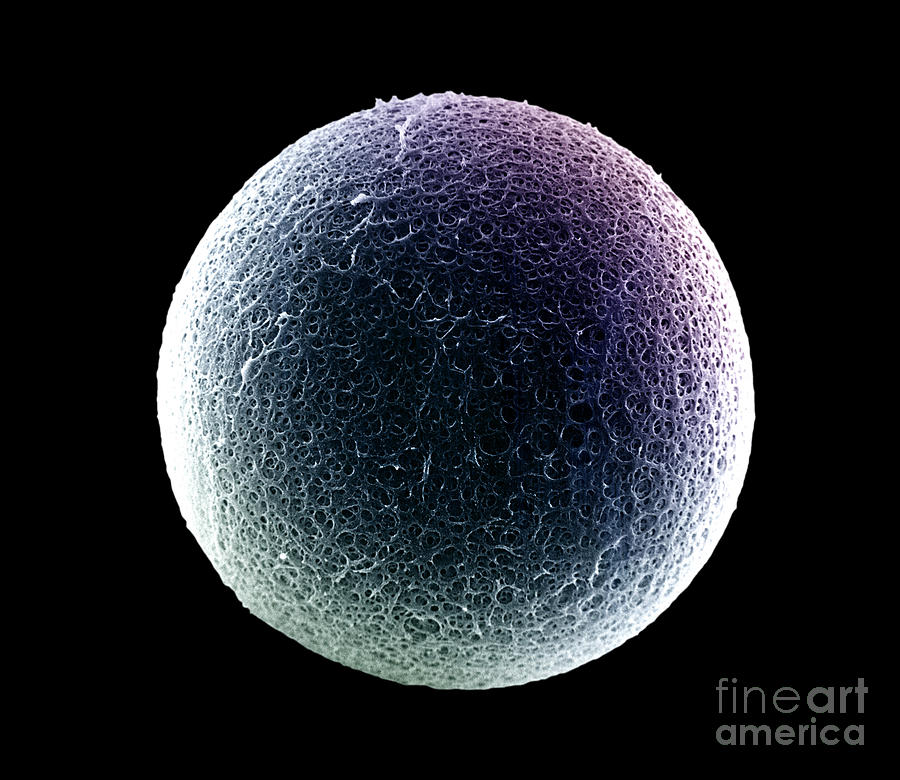
Sequence elements in the ZPD and the carboxy-terminal propeptide of ZP1–4 regulate secretion and assembly of nascent ZP proteins. Fibrils are oriented differently with respect to the oolemma in the inner and outer layers of the ZP. Nascent ZP proteins are synthesized, packaged into vesicles, secreted into the extracellular space, and assembled into long, crosslinked fibrils that have a structural repeat, a ZP2-ZP3 dimer, and constitute the ZP matrix. Genes encoding ZP proteins are conserved among mammals, and their expression is regulated by cis-acting sequences located close to the transcription start-site and by the same/similar trans-acting factors. Mouse and human ZP genes are encoded by single-copy genes located on different chromosomes and are highly expressed in the ovary by growing oocytes during late stages of oogenesis. Both subdomains adopt immunoglobulin (Ig)-like folds for their 3-dimensional structure. ZP1–4 have a large region of polypeptide, the ZP domain (ZPD), consisting of two subdomains, ZP-N and ZP-C, separated by a short linker region, that plays an essential role in polymerization of nascent ZP proteins into crosslinked fibrils. Unlike ECM surrounding somatic cells, the ZP is composed of only a few glycosylated proteins, ZP1–4, that are unique to oocytes and eggs. All mammalian oocytes and eggs are surrounded by a relatively thick extracellular matrix (ECM), the zona pellucida (ZP), that plays vital roles during oogenesis, fertilization, and preimplantation development.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
